Furthermore, removal of the template to obtain effective MIPs is also difficult. The overall performance of large and macroscopic bulk materials-based MIPs is compromised due to the heterogeneity of their analyte binding site and the poor accessibility of interior binding sites. MIPs can be fabricated in various formats such as films, membranes, hydrogels, microparticles, multi-walled carbon nanotubes (MWCNTs), or nanoparticles. Herewith, the polymer carries the memory that can selectively recognize the targeted molecule.ĭecades of research have demonstrated the superior performance of micro- and nanosized MIPs compared to their bulk macroscopic counterparts. After the polymerization, the template molecule is removed from the matrix to obtain the complimentary cavities. Then, cross-linkers arrest the temporary structure into a permanent one during the polymerization process to yield a three-dimensional polymeric network. The preparation of MIPs first requires the formation of a pre-assembly between the template molecule and functional monomers via either covalent or non-covalent interactions. Molecular imprinting is an approach that entails creating artificial recognition sites by polymerizing functional and cross-linking monomers in the presence of a target molecule ( Figure 1). New synthetic materials have recently been proposed to provide more stable, durable, and cheaper alternatives. 
However, using these biological components in clinical applications remains limited due to their physical and chemical instability and high cost. Nature employs biological receptors such as enzymes, antibodies, or histones as tools to recognize the respective antigens. 
Many diagnostic and therapeutic platforms require selective recognition of a target molecule. This review aims to provide a general overview of key recent advances in the fabrication of MIPs-coated inorganic nanoparticles and highlight their biomedical applications, including drug delivery, biosensor, bioimaging, and bioseparation. These materials with a multicomponent hierarchical structure composed of an inorganic core and an imprinted polymer shell exhibit enhanced properties and new functionalities. The integration of MIPs with inorganic nanomaterials such as silica (SiO 2), iron oxide (Fe 3O 4), gold (Au), silver (Ag), and quantum dots (QDs) combines several attributes from both components to yield highly multifunctional materials. In particular, MIP-coated inorganic nanoparticles have emerged as a promising platform for various biomedical applications ranging from drug delivery to bioimaging. Under 2-propanol conversions, the data seems to suggest that core-shell structure provides a synergistic effect for acetone production, however, this cannot be concluded until further testing is accomplished.Molecularly imprinted polymers (MIPs) continue to gain increasing attention as functional materials due to their unique characteristics such as higher stability, simple preparation, robustness, better binding capacity, and low cost. Under CO oxidation, the 1:4 structure calcined at 300 degrees Celsius presented the best results, and the 1:4 ratio was still active at 100 degrees Celsius after thermal treatments. The 1:4 gold-144 iron oxide catalyst produced the best catalytic activity and was further investigated using 2-propanol conversion as well as thermal treatments using CO oxidation. Using CO oxidation, each gold-144 iron oxide catalyst loaded onto silica gave varying degrees of full CO conversion depending on the thickness of the iron oxide layer. The gold-144 iron oxide particles with Au:Fe mass ratios of 1:2, 1:4, and 1:6 were synthesized and then deposited onto silica via colloidal deposition. Shell thickness is varied depending on the amount of iron precursor used and studied under the particle’s catalytic efficiency with carbon monoxide oxidation. 
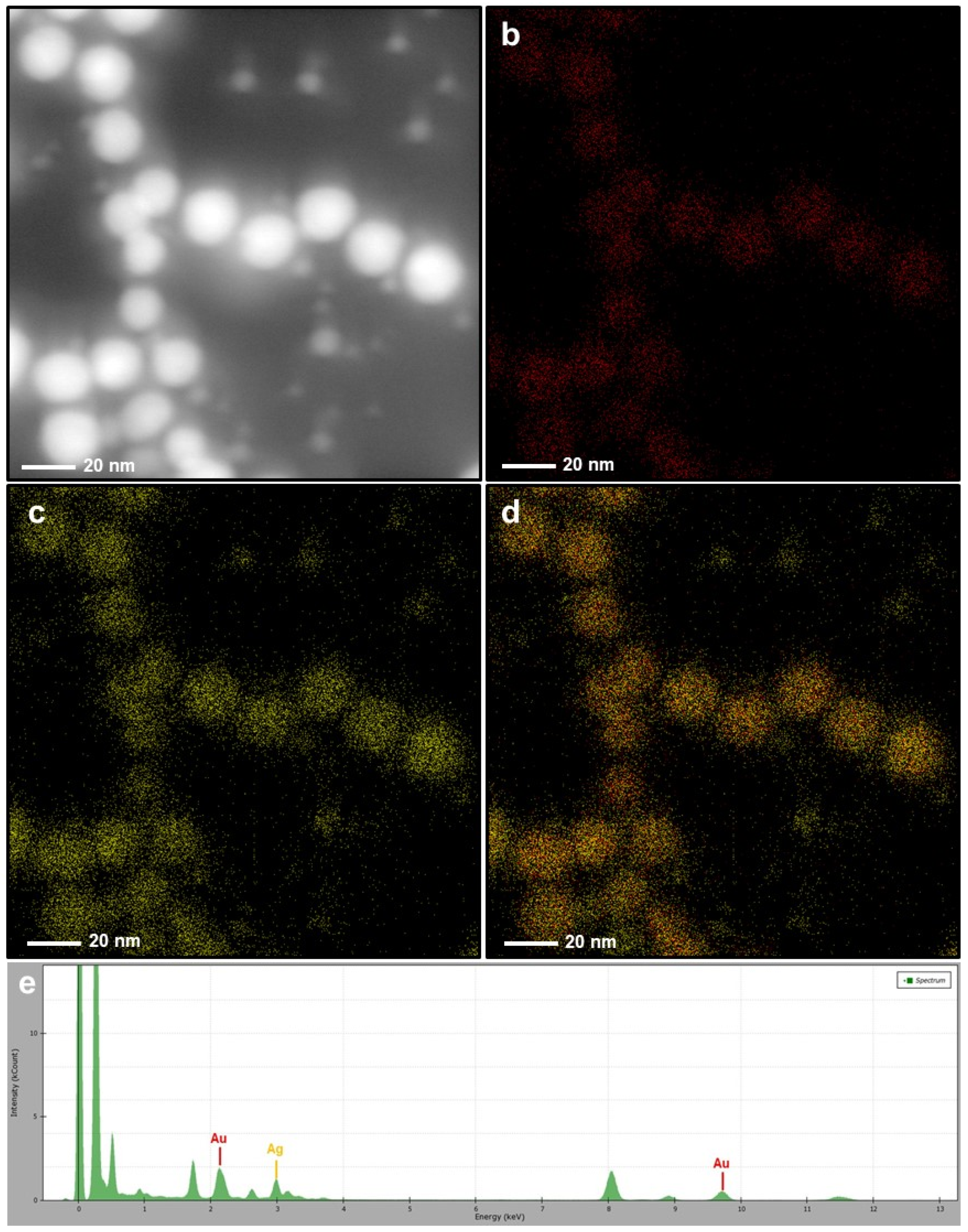
The research contained in this document discusses the synthetic procedure of a gold-144 cluster using a variation of the Brust-Schiffrin method followed by an iron oxide coating via post-selective oxidative treatment to create a gold-144 iron oxide core-shell structure. Core-shell heterostructures have been utilized as a catalyst that is thermally stable and exhibits a synergistic effect between core and shell, resulting in increased catalytic activity. Given the nature of small gold particles to sinter, it was found to be advantageous to protect the particles using a gold-metal oxide core-shell configuration. Thus, gold clusters are desirable given their uniformity, high surface-to-volume ratio, and high catalytic activity. This is even more so with gold nanoparticles in that gold only becomes catalytically active with small particles sizes. In the realm of catalysis, small nanoparticles have been an area of interest due to their high surface-to-volume ratio. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
